Learn the complete list of documents required for BIS certification in India including legal, technical, testing, factory, FMCS, and quality control documents for manufacturers and importers.
One of the most important parts of the BIS certification process is documentation. Whether applying for ISI certification, CRS registration, or FMCS approval, manufacturers are required to submit a wide range of technical, legal, manufacturing, and quality-related documents during the certification process.
In many cases, delays in BIS approval are not caused by testing failures or discrepancies during factory inspections, but by incomplete or inconsistent documentation.
Missing records, incorrect product descriptions, outdated calibration certificates, mismatched addresses, or improperly prepared technical documents can easily result in clarification rounds and additional processing time.
For manufacturers and importers unfamiliar with the BIS certification system, understanding the required documentation early can significantly improve preparation and reduce avoidable delays.
This guide explains the major categories of documents commonly required during the BIS certification process and highlights practical considerations manufacturers should keep in mind before applying.
Why Documentation Matters in BIS Certification
BIS certification is not only about product testing. The certification process is also intended to evaluate whether the manufacturer has the capability to consistently produce products conforming to the applicable Indian Standard.
Documentation helps BIS assess:
- manufacturing capability
- product consistency
- traceability systems
- testing infrastructure
- quality control procedures
- regulatory authenticity
Well-prepared documentation creates confidence during evaluation and inspection.
On the other hand, poor documentation often raises concerns regarding:
- operational readiness
- process control
- technical consistency
- quality system implementation
For this reason, manufacturers should treat documentation preparation as an important part of compliance readiness rather than merely an administrative formality.
Common Categories of Documents Required for BIS Certification
The exact documents required may vary depending on:
- certification scheme
- product category
- applicable Indian Standard
- manufacturing location
- product complexity
However, most applications generally involve the following categories:
- company and legal documents
- manufacturing documents
- quality control documents
- technical product documents
- laboratory and testing documents
- authorization and declaration documents
Understanding these categories makes the preparation process much easier.
Company and Legal Documents
Legal and company-related documents are required to establish the identity and legitimacy of the applicant.
These documents help verify:
- ownership
- factory location
- operational status
- legal registration
- authorized signatories
Company Incorporation Documents
Manufacturers are generally required to provide proof of company registration or legal establishment.
Depending on business structure, this may include:
- certificate of incorporation
- partnership deed
- proprietorship declaration
- LLP registration documents
The business name should remain consistent across all submitted documents.
Factory License
A valid factory license is commonly required for manufacturing units.
The details should match:
- manufacturing address
- company name
- production activities
Expired or inconsistent licenses often create complications during evaluation.
GST Registration Certificate
GST registration is frequently required as part of the application process.
Manufacturers should ensure:
- legal name consistency
- address accuracy
- active registration status
Minor inconsistencies between GST records and other documents can sometimes trigger clarification requests.
MSME Registration (If Applicable)
If the manufacturer is registered as an MSME, supporting documents may also be submitted.
Although not always mandatory, this may sometimes be useful for administrative purposes.
Import Export Code (IEC)
For importers and foreign trade-related activities, the Import Export Code issued by DGFT may also be required.
This becomes especially relevant in:
- FMCS applications
- importer-related compliance
- foreign manufacturer coordination
Manufacturing Related Documents
Manufacturing-related documents help BIS understand how the product is produced and whether the facility possesses adequate production capability.
These documents become particularly important during factory inspection and process evaluation.
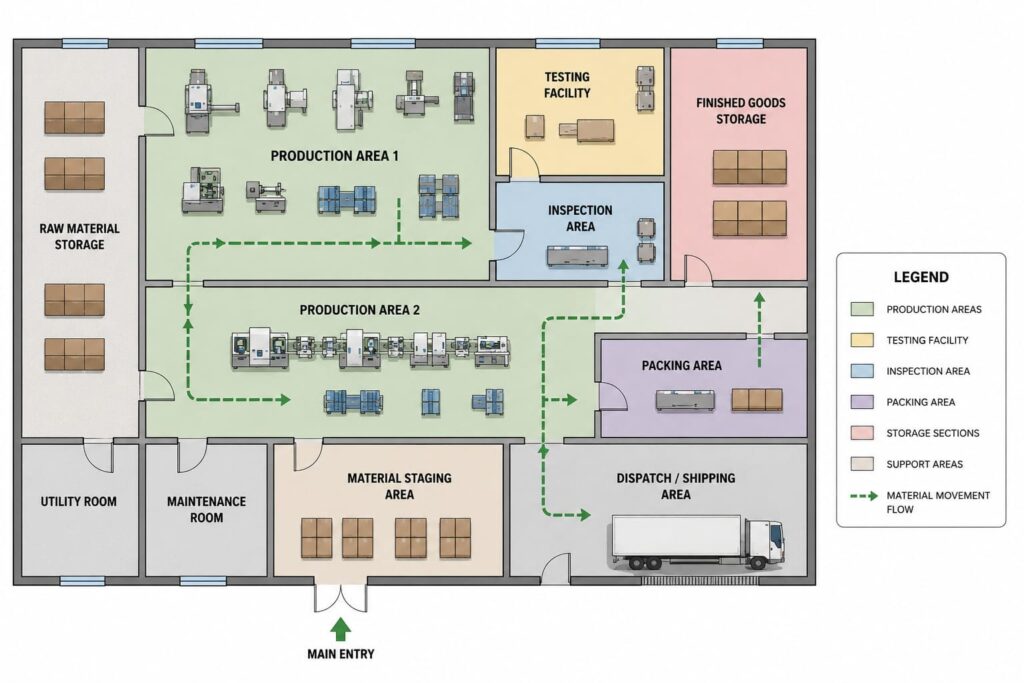
Factory Layout
Manufacturers are commonly required to provide a factory layout or plant layout showing:
- production areas
- storage sections
- testing facilities
- inspection areas
- material movement flow
A clear layout helps inspectors understand manufacturing organization and operational structure.
Manufacturing Process Flow Chart
A process flow chart explains how the product moves through various manufacturing stages.
This may include:
- raw material inspection
- production stages
- assembly
- in-process inspection
- final testing
- packing
- dispatch
Well-prepared process flowcharts demonstrate process clarity and operational control.
Machinery List
A detailed machinery list is often required.
This generally includes:
- machine names
- capacity
- quantity
- operational purpose
Inspectors may verify whether the listed machinery is actually available and operational during inspection.
Production Capability Details
Manufacturers may also need to provide:
- production capacity
- manufacturing capability
- shift operations
- manpower details
This helps BIS evaluate whether the unit is adequately equipped for consistent production.
Quality Control Documents
Quality control documentation is extremely important in BIS certification because conformity assessment depends heavily on process consistency and internal quality systems.
Manufacturers with organized quality systems usually experience smoother inspections and evaluations.
Quality Manual
A quality manual describes the company’s internal quality procedures and operational controls.
It may include:
- inspection systems
- process monitoring
- corrective action procedures
- quality responsibilities
- testing controls
Even small manufacturing units benefit significantly from maintaining structured quality documentation.
Test Equipment List and Calibration Certificates
A list of Test Equipment available at your in-house testing lab is also required to be submitted. Calibration certificates of the test equipments are among the most commonly verified documents during inspection.
Testing equipment used for quality control should generally be:
- calibrated
- traceable
- valid
- properly maintained
Expired calibration certificates are one of the most common inspection observations in many factories.
Manufacturers should ensure calibration validity before scheduling inspection.
Internal Test Records
Factories are often expected to maintain internal testing records demonstrating routine quality checks.
These records may include:
- raw material inspection reports
- in-process test records
- final inspection reports
- quality check logs
Properly maintained records help establish manufacturing consistency.
Standard Operating Procedures (SOPs)
Some manufacturers maintain documented procedures for:
- production operations
- testing activities
- handling procedures
- quality checks
These SOPs help standardize manufacturing activities and reduce operational inconsistency.
Product Technical Documents
Technical documentation helps BIS understand the product itself and verify conformity with the applicable Indian Standard.
The accuracy of technical documents is extremely important.
Product Specifications
Detailed product specifications are generally required.
These may include:
- dimensions
- ratings
- material specifications
- technical parameters
- operating characteristics
The specifications should match:
- test reports
- labeling
- product markings
- application forms
Inconsistencies are a common source of delays.
Product Drawings
Technical drawings may be required for many products.
These drawings help demonstrate:
- construction details
- assembly configuration
- dimensional requirements
- component arrangement
Clear technical drawings improve evaluation efficiency.
Bill of Materials (BOM)
For certain products, manufacturers may need to provide a Bill of Materials listing:
- components
- subassemblies
- raw materials
- specifications
This becomes especially important for electronics and engineered products.
Product Labels and Marking Artwork
Manufacturers may need to submit:
- label artwork
- packaging details
- marking layouts
These are reviewed to ensure proper compliance with applicable marking requirements.
Incorrect labeling is a common compliance issue in many industries.
Product Manuals and Product Literature
Product manuals are made available for easy access by all. The Product manual guides you on what tests are to be conducted and what frequency is prescribed. You will also be required to submit an acceptance to the prescribed Scheme of Inspection and Testing as mentioned in the product manual.
Alternatively, you can also submit your proposal to follow a different testing frequency and undertake to maintain your declared proposals.
Test Reports
Test reports are one of the most critical components of the BIS certification process. In-house test report of the varieties you intend to get it convered in the scope of your license is mandatory.
The reports demonstrate whether the product complies with the applicable Indian Standard.
Laboratory Recognition
Testing is generally required through:
- BIS laboratories
or - BIS-recognized laboratories
Manufacturers should ensure the selected laboratory is authorized for the applicable standard and product category.
Validity of Test Reports
Manufacturers should ensure:
- reports are recent
- reports are complete
- all required parameters are covered
- product descriptions match the application
Expired or incomplete reports frequently create delays.
Sample Consistency
The tested sample should accurately represent the actual production product.
Differences between:
- tested sample
- commercial production
- product labeling
- technical specifications
can create serious compliance concerns later.
Documents Required for Foreign Manufacturers
Foreign manufacturers applying under schemes such as FMCS often require additional documentation.
These documents help establish:
- overseas manufacturing legitimacy
- local representation
- compliance accountability
Authorized Indian Representative (AIR) Documents
Foreign manufacturers may need to appoint an Authorized Indian Representative.
Documents may include:
- nomination letters
- authorization agreements
- identity documents
- address proof
The AIR acts as the local compliance representative in India.
Overseas Manufacturing Licenses
Foreign factories may also need to provide:
- business licenses
- factory registrations
- manufacturing approvals
- export-related documents
These documents help establish manufacturing authenticity.
Trademark Authorization (If Applicable)
Where branding and manufacturing entities differ, trademark authorization documents may also be required.
This is common in:
- OEM manufacturing
- private labeling arrangements
- contract manufacturing models
Common Documentation Mistakes
Documentation errors are one of the most common causes of delay in BIS certification.
Many issues arise not because documents are missing entirely, but because the submitted information is inconsistent across different records.
Inconsistent Product Descriptions
Manufacturers often use slightly different product names across:
- test reports
- applications
- labels
- technical documents
Even small inconsistencies can trigger clarification requests.
Maintaining standardized product descriptions is extremely important.
Address Mismatches
Factory addresses should remain consistent across:
- licenses
- GST records
- application forms
- calibration certificates
Address inconsistencies are surprisingly common in certification applications.
Expired Calibration Certificates
Manufacturers sometimes overlook calibration validity before inspection.
Expired calibration records immediately create concerns regarding testing reliability.
Calibration schedules should always be reviewed in advance.
Incomplete Technical Documentation
Missing:
- drawings
- specifications
- labeling artwork
- component information
often delays technical evaluation.
Organized documentation preparation significantly improves processing efficiency.
Tips for Faster BIS Documentation Preparation
Proper planning can make the documentation process much smoother.
Start Documentation Early
Many businesses begin preparing documents only after testing has started.
This often creates avoidable delays.
It is much better to prepare:
- legal records
- technical documents
- calibration certificates
- quality manuals
well before formal application.
Maintain Digital Records
Maintaining organized digital copies helps:
- reduce confusion
- speed up submission
- simplify revisions
- improve traceability
Structured documentation systems become increasingly valuable as compliance activities grow.
Review Consistency Carefully
Before submission, manufacturers should verify:
- names
- addresses
- model numbers
- technical ratings
- labeling details
across all documents.
This simple review process prevents many common issues.
Prepare for Inspection Practically
Documentation alone is not enough.
Factories should ensure:
- actual processes match submitted records
- testing equipment is operational
- personnel understand procedures
- records are properly maintained
Inspection readiness should reflect actual operational practices.
Frequently Asked Questions (FAQs)
Are the documents required for BIS Certification the same for all schemes?
No. Documentation requirements vary depending on:
- ISI certification
- CRS registration
- FMCS certification
- product category
- applicable standard
Is factory inspection mandatory for all products?
Not always. Inspection requirements depend on the certification scheme and product category.
Can expired test reports be used for BIS certification?
Generally, manufacturers should use valid and recent test reports. Expired reports may not be accepted.
Why are calibration certificates important?
Calibration certificates help establish the reliability and accuracy of testing equipment used during quality control activities.
Do foreign manufacturers require additional documents?
Yes. Foreign manufacturers often need additional documentation related to:
- AIR appointment
- overseas manufacturing registration
- legal authorization
- factory details
Final Thoughts
Documentation plays a major role in the BIS certification process.
Well-prepared documentation not only improves processing efficiency but also reflects the manufacturer’s operational discipline and compliance readiness.
Manufacturers that maintain:
- organized records
- structured quality systems
- accurate technical documentation
- valid calibration systems
generally experience smoother certification workflows and fewer delays.
As regulatory compliance in India continues expanding across industries, proper documentation management is becoming an increasingly important part of successful manufacturing and import operations.

